A new perspective on reactive oxygen species
Reactive oxygen species (ROS), often referred to as free radicals, have a bad reputation – and for good reason. At high levels, these reactive forms of oxygen, which have unpaired electrons in their outer orbits, can cause significant cellular damage. For example, they destabilize cell membranes, render proteins nonfunctional and mutate DNA, which then can lead to cancer. Concerned about these issues, some people take antioxidants, which eliminate free radicals by donating electrons, to protect themselves against damage from ROS.
Despite clear evidence that high levels of ROS have adverse health effects, studies in recent years have also begun to demonstrate that these molecules have normal physiological roles as well. In a new study published in the journal Molecular Cell, investigators from the Joslin Diabetes Center and Harvard Medical School discovered that ROS can play a role in increasing a cell’s resistance to stress. In their research, the scientists used the DCP-Bio1 protein cysteine oxidation probe, which is made available on the Kerafast platform by the laboratory of Dr. Leslie B. Poole at the Wake Forest School of Medicine.
One protein, two functions
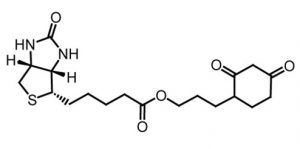
The DCP-Bio1 protein cysteine oxidation probe, used in a new Molecular Cell study on reactive oxygen species.
The research team studied a protein called IRE-1, a protein kinase in the outer membrane of the endoplasmic reticulum (ER), the cell organelle responsible for synthesizing proteins and lipids. IRE-1’s typical role is to detect misfolded proteins and turn on a corrective mechanism called the unfolded protein response (UPR). The protein has long been studied for its impact on ER health and disease.
In the new study, the scientists looked at IRE-1 in both the model system C. elegans and human cells and discovered another role for the protein: triggering an antioxidant response that boosts cellular stress resistance and lifespan. When IRE-1 gets a signal from ROS, the unfolded protein response is inhibited and the antioxidant response to initiated. This change in function, termed a “functional switch” by the researchers, is caused by a single oxygen molecule sulfenylating a single cysteine.
“It’s surprising to see the same molecular sensor doing two different things, like a combination smoke/carbon monoxide alarm,” senior author Dr. Keith Blackwell said in a press release. “A major sensor of misfolded proteins in the ER turns out to be a major sensor of ROS, acting in a completely different molecular pathway with a completely different function.”
Takeaways and next steps
The researchers are now exploring how IRE’s ROS and ER pathways could be targeted for drug development in diseases such as diabetes. One area of interest is how ROS molecules influence gene regulatory responses in cells exposed to signals such as elevated glucose levels. For now, one takeaway the researchers point out is that excessive antioxidant use might do more harm than good.
“In clinical trials, therapies with antioxidants have been pretty much a bust, and it’s not entirely clear why,” Dr. Blackwell said. “But blindly consuming large doses of antioxidants is probably not the best idea, because while your intent would be to protect yourself from damage, you’re also potentially interfering with normal ROS signals that are helpful and important.”
If you work in this area of research, check out our reagents for redox biology research, including a variety of unique cysteine oxidation probes. You may also be interested in our rough endoplasmic reticulum antibody, endoplasmic reticulum-related services or reagents for diabetes research.


