4 Recent Advances in Click Chemistry
Biomolecule labeling requires reaction procedures that can be performed under physiological conditions (e.g., neutral pH, aqueous solution, ambient temperature) with low reactant concentrations to ensure non-toxic, low background labeling at reasonable time scales while still preserving biological function. Click chemistry can be an excellent method to accomplish this. Below, we explain how traditional copper-catalyzed click chemistry works; you can also check out our Click Chemistry 101 blog post to learn how the method is used for applications such as creating fluorescent antibodies and detecting sulfenic acid. Then, we introduce four fairly new bio-conjugation methods that each offer various improvements over traditional click chemistry.
Traditional Copper-Catalyzed Click Chemistry
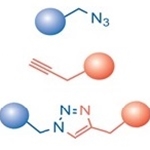
The most prominent example of click chemistry is the copper-catalyzed azide-alkyne click chemistry reaction. A terminal alkyne-functionalized molecule reacts with an azide-functionalized molecule to form a stable conjugate via a triazole moiety.
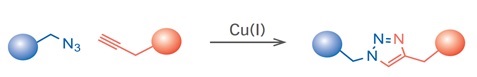
Because terminal alkynes are fairly unreactive towards azides, the efficiency of this type of reaction strongly depends on the presence of a metal catalyst such as copper (Cu). However, the use of copper-catalyzed click chemistry reactions in live cells can be difficult due to the toxicity of Cu(I) ions.
Features:
- Small-sized azides and alkynes possess excellent substrate properties
- Optimization of assay conditions required (type and concentration of copper source, reduction reagent and copper ligand)
- Suitable if potential copper toxicity does not matter (not recommended for in vivo or live cell labeling)
Dibenzylcyclooctyne (DBCO)-Based Click Chemistry
A copper-free and non-toxic alternative labeling method of azides utilizes strained cyclooctynes such as dibenzylcyclooctyne (DBCO). DBCO possesses a remarkably decreased activation energy in contrast to terminal alkynes and thus does not require an exogenous catalyst. DBCO-based reagents combine high reactivity with sufficient hydrophility, allowing for low background labeling of azide-functionalized molecules with even greater efficiency than copper-catalyzed click chemistry methods.
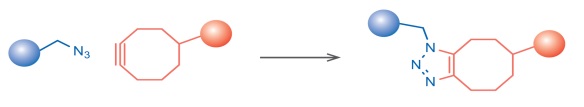
Features:
- Biocompatible reaction occurs under mild aqueous buffer conditions
- No catalyst or accessory reagents required
- Faster detection of small-sized azides compared to traditional, copper-catalyzed click chemistry reactions
- Suitable for dual-labeling approaches in combination with tetrazine – trans-cyclooctene ligation
Tetrazine – trans-Cyclooctene (TCO) Ligation
Tetrazine – trans-cyclooctene (TCO) ligation constitutes a non-toxic biomolecule labeling method of unparalleled speed that is ideally suited for in vivo cell labeling and low concentration applications. A tetrazine-functionalized molecule (A) reacts with a strained (e.g., DBCO) alkene-functionalized molecule (B) thereby forming a stable conjugate (A-B) via a dihydropyrazine moiety.

Features:
- Extremely fast kinetics (> 800 M -1s-1) permits efficient protein-protein conjugation at < 1 mg/mL in 60 minutes or less
- Biocompatible reaction occurs under mild aqueous buffer conditions at ambient temperature
- No toxic catalyst or accessory reagents required
- Suitable for dual-labeling approaches in combination with the strain-promoted azide – DBCO reactions
Copper Chelating Click Chemistry
In copper chelating click chemistry, a copper-chelating group has been incorporated to an azide structure to allow co-diffusion and concentrate copper at the reaction site. The picolyl azide effect is so strong that it allows a copper catalyst’s concentration to be decreased from 100 μm to 10 μm without any adverse effect on the reaction.
Features:
- Reduce reaction toxicity by omitting THPTA ligand or decreasing copper concentration 10-fold, allowing for use of standard copper-catalyzed click chemistry probes
- When used in traditional copper-catalyzed click chemistry conditions, efficiency is dramatically increased
Bis-sulfone Conjugation Reagents
Traditional sulfhydryl labeling chemistry cleaves disulfide bridges and leaves them in “open form”, which can compromise the stability and function of an antibody or other proteins. To solve this issue, bis-sulfone reagents are used to re-bridge the disulfide bond, thus enhancing the structural stability of the protein. The approach uses bis-sulfone reagents that are selective for the cysteine sulfur atoms from a native disulfide that has been reduced.
Features:
- Overcomes the drawbacks of traditional sulfhydryl labeling chemistry
- ThioLinker bis-sulfone reagents add the advantage of being water-soluble, greatly improving not only solubility, but also labeling and re-bridging efficiency
In summary, click chemistry has become a widely used and versatile tool for many different researchers. The method works rapidly at mild aqueous conditions and provides high yields. Yet despite these benefits, traditional click chemistry has several drawbacks, including its use of copper as a catalyst, which limits in vivo and live cell applications. Researchers have therefore begun to develop improved click chemistry reagents that offer advantages such as biocompatibility and increased efficiency. To see if any available reagents might help advance your research, check out our full list of click chemistry materials and contact TechnicalSupport@kerafast.com with any questions.