Intro to Adeno & Lentiviruses
In nature, adenoviruses and lentiviruses can be dangerous – HIV is a lentivirus, and adenoviruses can cause ailments such as the common cold, bronchitis and pneumonia. But in research, the two viruses are extremely useful, serving as powerful gene delivery tools that can introduce genes of interest into target cells. Here you’ll find an introduction to adenoviruses and lentiviruses, as well as several quick tips for successful transduction.
A key difference between the two types of viruses is that adenoviral DNA does not integrate into the host cell genome and thus is not replicated during cell division. Lentiviral DNA does get incorporated into the host cell genome and is replicated along with the cell.
Both types of viruses are highly applicable to research as a gene delivery tools. You start with a target vector – which contains the gene to be introduced – and a packaging vector, which contains the genes encoding necessary virion proteins. You then co-transfect 293T cells with these vectors. The cells produce the virus, after which you collect, concentrate and purify the viral particles. Finally, you transduce the virus into target cells, which express the gene of interest.
Tips for retrovirus transduction
There are several things to keep in mind when working with retroviruses to help ensure successful transduction into target cells. First, it is important to rely on a functional titer rather than a non-functional (or physical) titer, if possible. Non-functional titer is a measure of the amount of virus present, typically calculated using methods such as qPCR or P24 quantitation by ELISA. These methods are simple, yet they often overestimate infectivity.
In contrast, functional titer measures how much of the virus present can actually infect cells, expressed in transduction units per milliliter (TU/mL). This is a more accurate value, though it is more time-consuming and not always feasible to calculate. Common methods for determining functional titer include counting the colony forming units after antibiotic selection and using flow cytometry if a fluorescent protein is being expressed.
In addition, to achieve greater transduction, simply using more volume of the virus may not be the best idea. Packaging supernatants can sometimes inhibit transduction and be toxic to cells. Rather, it is often best to use a more concentrated virus. This allows for a higher multiplicity of infection (MOI), or the ratio of infection agents to targets (i.e., the number of virus particles divided by the number of host cells). As the MOI increases, the percentage of host cells infected with at least one viral particle also increases.
With successful transduction, retroviruses are useful for a wide variety of applications, including:
- Expressing bright fluorescent proteins for whole cell tracking
- Expressing tagged fluorescent proteins for organelle tracking
- Expressing Tet-On (tetracyclicine inducible) transactivtor to regulate transcription
- Expressing iPS factors for reprograming
Premade and custom Adenoviruses and Lentiviruses
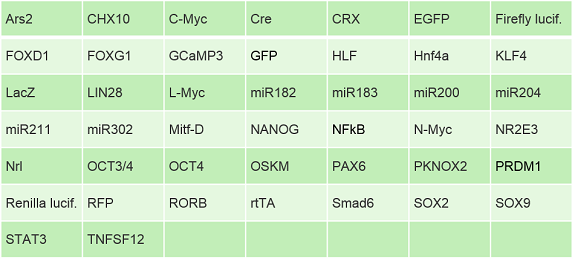
If creating your own viruses sounds too time-consuming or labor-intensive, Kerafast can provide a variety of premade adenoviruses and lentiviruses. Adenovirus offerings include the following premade targets, and custom constructs can be produced in as little as three weeks.
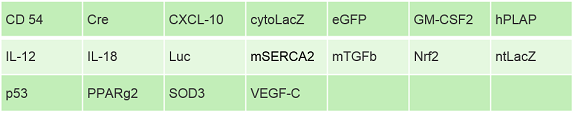
We also supply premade lentiviruses ready-to-use for both in vitro and in vivo transduction. These lentivirus particles offer high functional titers (in fact, their functional titers have been found to be 10x greater than other commercially available lentiviruses) and small “single-use” aliquot sizes that avoid the titer loss that often occurs with repeat freeze-thaw cycles. The lentiviruses are also highly pure and serum-free, containing no contaminants that could interfere with transduction efficiency.
The following premade targets are accessible on the Kerafast site. In addition, a custom lentivirus service with quick turnaround times is available for any other needs.