New study suggests patient’s own skin cells may target glioblastoma
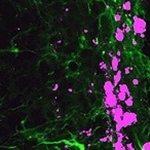
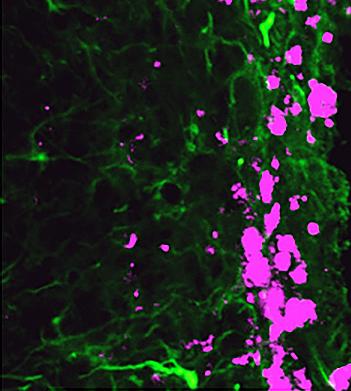 Reprogrammed stem cells (green) chase down and kill glioblastoma cells (pink), potentially offering a new and more effective treatment option for a disease that has not had any in more than 30 years. |
A recent study by researchers at the University of North Carolina at Chapel Hill has identified a potential new treatment for glioblastoma. Glioblastoma represents approximately 15% of brain tumors and is one of the most aggressive types of brain cancer. Only about 5% of those diagnosed survive more than five years.
Dr. Shawn Hingtgen led the study published this month in Nature Communications, which utilized mouse fibroblast cells, a type of skin cell. Using a technique for which Dr. John B. Gurdon and Dr. Shinya Yamanaka won the Noble Prize in Medicine in 2012, Hingtgen’s laboratory reprogrammed the fibroblast cells via transdifferentiation to become induced neuronal stem cells (iNSC). This technique essentially takes development backward, where differentiated cells return to a pluripotent state. Using mouse glioblastoma tumor models, the researchers found that the iNSCs were actually tumorigenic and migrated to the site of tumors. Further, the team engineered the iNSCs to produce tumor necrosis factor-α-related apoptosis-inducing ligand, a molecule that causes apoptosis, or cell death. The reprogramed iNSCs demonstrated that they were excellent vehicles for targeting the glioblastoma and delivering the anti-cancer compounds. In fact, the study demonstrated a decrease of the growth of established solid and diffuse patient-derived orthotopic glioblastoma xenografts 230- and 20-fold, respectively. Furthermore, the survival time of the mice increased 160 to 220%. According to Dr. Hingtgen, “This is the first time this direct reprogramming technology has been used to treat cancer.” |
Now the researchers are finding ways to keep the stem cells organized and in the vicinity of the cancer cells long enough to kill any remaining cancerous tissue after surgical removal of the tumor, since current surgical procedures often are unable to remove the entire tumor effectively and the cancer returns. To this end, they are using a type of surgical glue which acts as a physical matrix to keep the cancer-fighting stem cells in the region up to three times longer. This research paves the way for a personalized medicine approach to treating glioblastoma and other cancers.
Do you work in this area of research? Check out our related reagents for cancer and stem cell research, including:
- U-251 MG Glioblastoma Cell Line from the Dr. Darell D. Bigner lab at Duke University
- Highly Systemic Metastatic Cell Lines from the Dr. Thomas N. Seyfried lab at Boston College
- Tumor Necrosis Factor Antibodies from the Royal A. McGraw lab at University of Georgia
- Various Premade Lentivirus Particles for the transformation of induced pluripotent stem cells